A Basic Guide To The Electronic Common Technical Document

Electronic Common Technical Document Ectd Pdf Food And Drug Per the fda data standards catalog, the electronic submission of ectd v4.0 to cber and cder is supported for new nda, bla, anda, ind, and mfs beginning september 16, 2024. only new applications. Learn the basics of the electronic common technical document and how pharmaceutical companies should familiarize themselves with the documents within it.
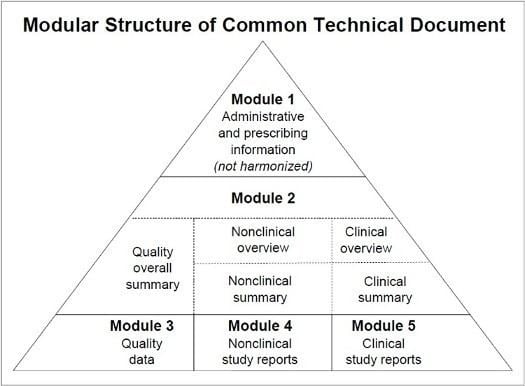
Electronic Common Technical Document Alchetron The Free Social The electronic common technical document (ectd) is a standardized, electronic format used to submit regulatory information about medicinal products to health authorities. the ectd has been developed under the guidelines of the international council for harmonisation (ich). The latest version of the ectd v4.0 implementation guide (v1.6) and ectd v4.0 controlled vocabulary package (v1.0.2) documents were endorsed by the ich assembly in may 2024. Designed for pharma professionals and clinical trial experts, this guide covers folder structures, lifecycle sequences, hyperlinks, metadata, and validation steps necessary for successful dossier delivery. Learn about electronic common technical document (ectd) for streamlined pharmaceutical regulatory submissions and global compliance.

What Is An Electronic Common Technical Document Ectd Designed for pharma professionals and clinical trial experts, this guide covers folder structures, lifecycle sequences, hyperlinks, metadata, and validation steps necessary for successful dossier delivery. Learn about electronic common technical document (ectd) for streamlined pharmaceutical regulatory submissions and global compliance. One of the most widely accepted standards for submitting regulatory documentation is the electronic common technical document (ectd). this globally recognized format simplifies submissions to regulatory bodies and improves the efficiency of review processes. The pharmaceutical industry has transitioned rapidly from paper based submissions to digital formats like the electronic common technical document (ectd). this shift streamlines global regulatory processes, supports lifecycle management, and aligns with digital transformation trends across agencies. This document serves as the practical guidance next to the eu implementation guide (ig) of the electronic common technical document (ectd) v4.0 using the hl7 version 3 regulated product submission (rps) release 2 normative for human medicinal products. In this guide, we’ll walk you through what ectd is, how it has evolved, the significance of version 4.0, its structure and components, and how sponsors can prepare for effective submission and lifecycle management.

Common Technical Document Vs Electronic Common Technical Document Pptx One of the most widely accepted standards for submitting regulatory documentation is the electronic common technical document (ectd). this globally recognized format simplifies submissions to regulatory bodies and improves the efficiency of review processes. The pharmaceutical industry has transitioned rapidly from paper based submissions to digital formats like the electronic common technical document (ectd). this shift streamlines global regulatory processes, supports lifecycle management, and aligns with digital transformation trends across agencies. This document serves as the practical guidance next to the eu implementation guide (ig) of the electronic common technical document (ectd) v4.0 using the hl7 version 3 regulated product submission (rps) release 2 normative for human medicinal products. In this guide, we’ll walk you through what ectd is, how it has evolved, the significance of version 4.0, its structure and components, and how sponsors can prepare for effective submission and lifecycle management.

Common Technical Document Vs Electronic Common Technical Document Pptx This document serves as the practical guidance next to the eu implementation guide (ig) of the electronic common technical document (ectd) v4.0 using the hl7 version 3 regulated product submission (rps) release 2 normative for human medicinal products. In this guide, we’ll walk you through what ectd is, how it has evolved, the significance of version 4.0, its structure and components, and how sponsors can prepare for effective submission and lifecycle management.

Common Technical Document Vs Electronic Common Technical Document Pptx
Comments are closed.