5 Common Missing Actions When Documenting A Defect

Defect Steps Pdf Operating System System We describe for you the 5 common missing actions when documenting a defect. This article explores the most common documentation pitfalls and how to build a robust system that supports safety rather than undermines it.

23 Defect Recording Pdf Information Technology Management Business From incomplete field updates to missing hidden elements, these as built documentation mistakes lead to costly rework. learn how to avoid them. The qa testing channel sdneorspot8r1m15e26he7e08310,dt10mti760c927b0iti3729lg · 5 common missing actions when documenting a defect 5 common missing actions when documenting a defect. Mistakes and failures are possible to happen in quality inspection. let’s figure out the common inspection failures, as well as the causes and methods to solve them. By identifying and fixing common mistakes, you can make your documentation clear, concise, and actually useful. in this article, we’ll go through five common documentation mistakes and.

Best Practices For Documenting And Reporting Defect Detection Mistakes and failures are possible to happen in quality inspection. let’s figure out the common inspection failures, as well as the causes and methods to solve them. By identifying and fixing common mistakes, you can make your documentation clear, concise, and actually useful. in this article, we’ll go through five common documentation mistakes and. Common causes of delays in construction projects include poor communication between stakeholders, unclear roles and responsibilities, inaccurate or incomplete documentation, and unforeseen issues like weather disruptions or supply chain delays. By being aware of these common mistakes, organizations can take proactive steps to avoid them, ensuring a more effective and compliant approach to managing nonconformities and corrective actions under iso 9001 clause 10.2. Recognizing these common pitfalls is the first step in fixing them. manual errors and inconsistencies. handwritten notes, lost papers, or rush entries are all breeding grounds for errors. while documentation is so dependent on manual input, errors are sure to occur. A common documentation failure is the “superficial” investigation. this happens when a firm fails to document a thorough root cause analysis. if your record simply states “human error,” the fda will view it as inadequate. they expect more than just “retraining” as a corrective action. a compliant record provides a transparent narrative.

Common Defect Management Mistakes Project Manaverse Common causes of delays in construction projects include poor communication between stakeholders, unclear roles and responsibilities, inaccurate or incomplete documentation, and unforeseen issues like weather disruptions or supply chain delays. By being aware of these common mistakes, organizations can take proactive steps to avoid them, ensuring a more effective and compliant approach to managing nonconformities and corrective actions under iso 9001 clause 10.2. Recognizing these common pitfalls is the first step in fixing them. manual errors and inconsistencies. handwritten notes, lost papers, or rush entries are all breeding grounds for errors. while documentation is so dependent on manual input, errors are sure to occur. A common documentation failure is the “superficial” investigation. this happens when a firm fails to document a thorough root cause analysis. if your record simply states “human error,” the fda will view it as inadequate. they expect more than just “retraining” as a corrective action. a compliant record provides a transparent narrative.
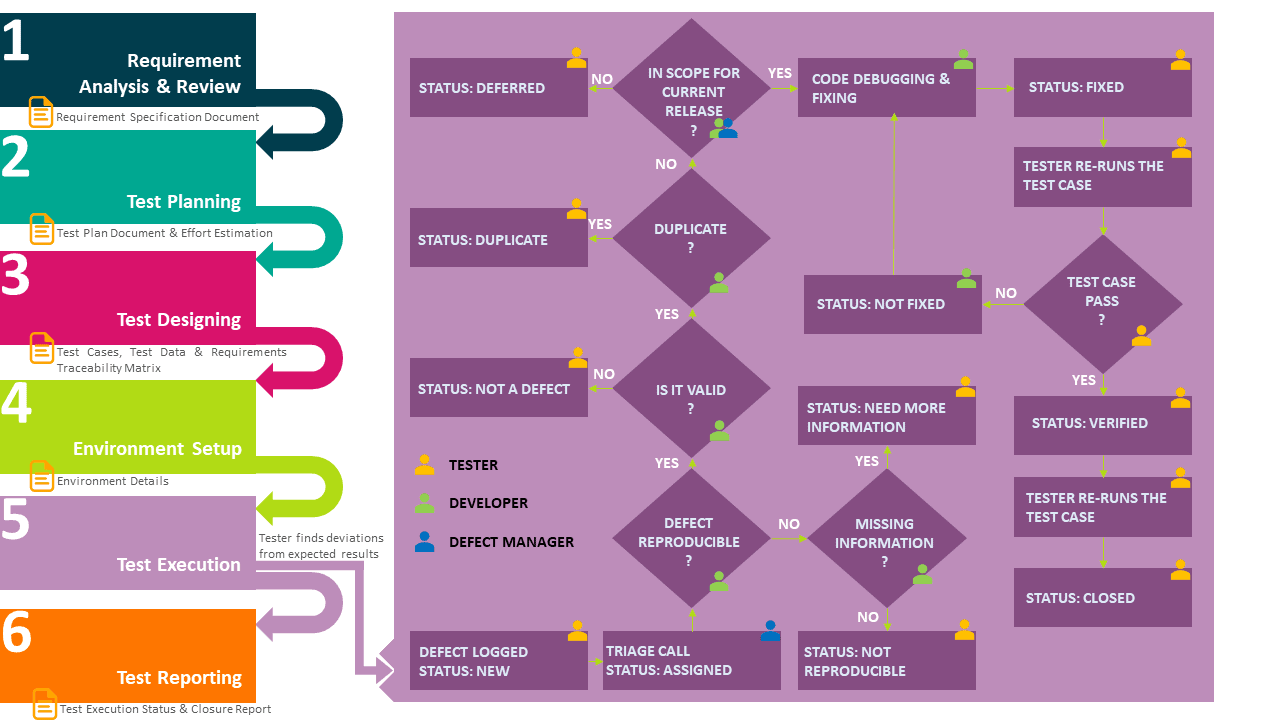
Defect Recognizing these common pitfalls is the first step in fixing them. manual errors and inconsistencies. handwritten notes, lost papers, or rush entries are all breeding grounds for errors. while documentation is so dependent on manual input, errors are sure to occur. A common documentation failure is the “superficial” investigation. this happens when a firm fails to document a thorough root cause analysis. if your record simply states “human error,” the fda will view it as inadequate. they expect more than just “retraining” as a corrective action. a compliant record provides a transparent narrative.
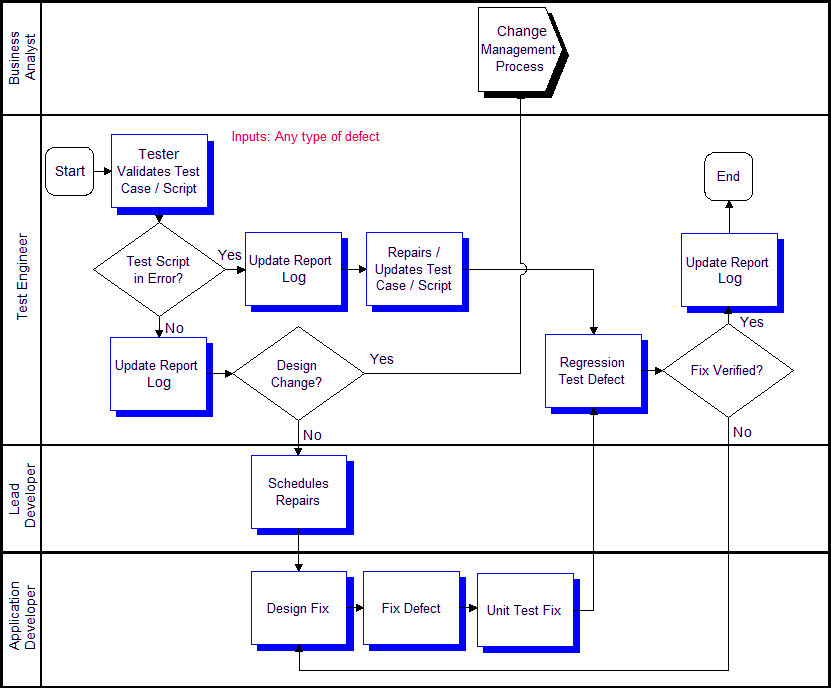
Defect Process Fasbuild
Comments are closed.