3 Yroven Oxysize

Malenkie Jenshini Yroven 3 Alcott Louisa May 9785171327781 Amazon Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on . Ozone (o₃) is a type of gas found in the earth’s atmosphere, made up of three oxygen atoms linked together. unlike the oxygen we breathe, which has two oxygen atoms, ozone has an extra oxygen atom that makes it quite special.
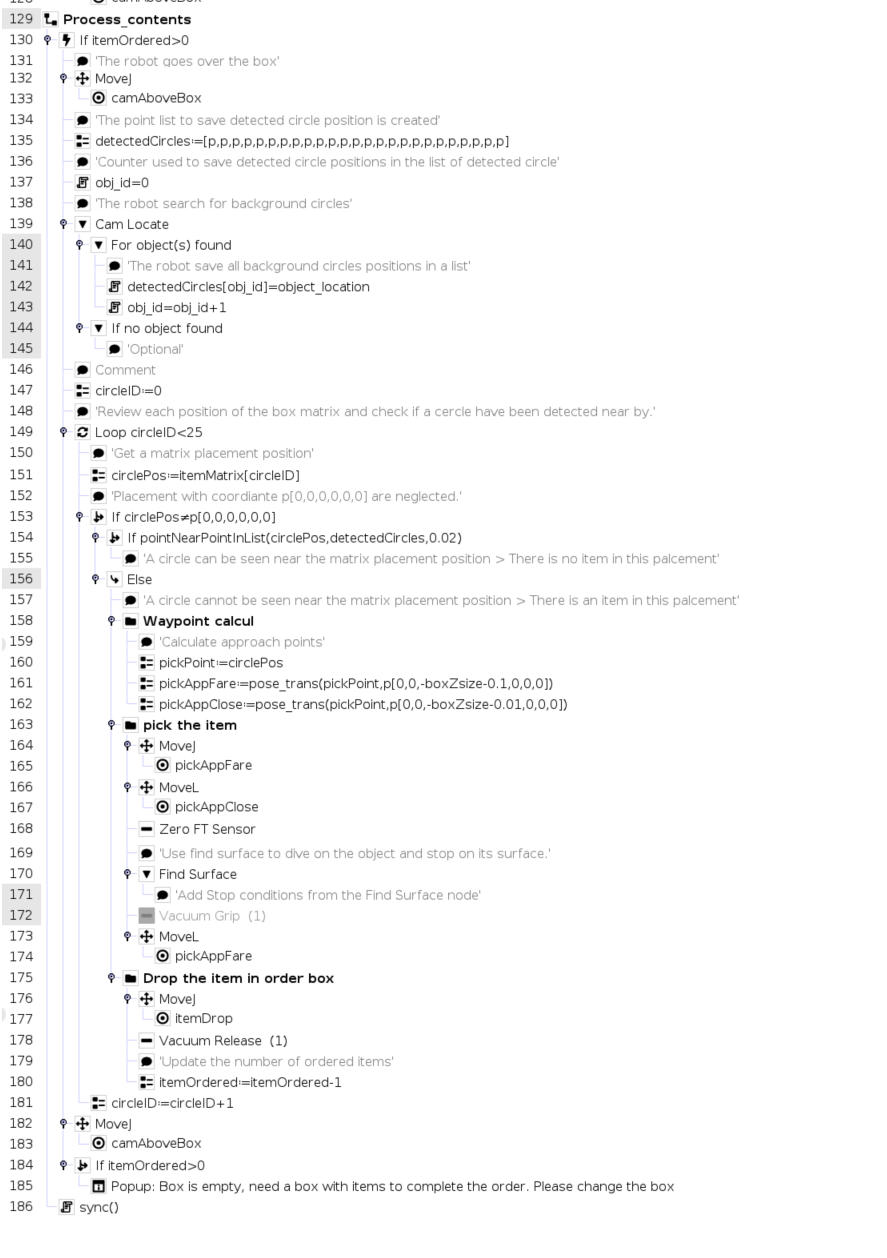
Order Preparation With Robotiq Wrist Camera Ozone (o3) is a molecule in which three atoms of oxygen are bonded together. the oxygen gas in the air we breathe has two oxygen atoms bonded together (o 2). ozone is relatively unstable and releases its third oxygen atom readily so it oxidizes and burns things more readily than oxygen gas. Ozone is a gas made up of three oxygen atoms (o 3). it occurs naturally in small (trace) amounts in the upper atmosphere (the stratosphere). ozone protects life on earth from the sun’s ultraviolet (uv) radiation. Ozone (o3) is defined as a strong oxidizing agent that can act as a biocide and readily decomposes into oxygen (o2) and oxygen free radicals, which react with bacterial cell contents. how useful is this definition? you might find these chapters and articles relevant to this topic. Ozone (o 3) is a molecule composed of three oxygen atoms, known for its strong oxidizing properties and its role in the earth’s upper atmosphere as a protective layer, absorbing the majority of the sun’s harmful ultraviolet radiation.

Ege Matematika Fynkcii Zadannie Grafikami I Ih Proizvodnie Zadacha Ozone (o3) is defined as a strong oxidizing agent that can act as a biocide and readily decomposes into oxygen (o2) and oxygen free radicals, which react with bacterial cell contents. how useful is this definition? you might find these chapters and articles relevant to this topic. Ozone (o 3) is a molecule composed of three oxygen atoms, known for its strong oxidizing properties and its role in the earth’s upper atmosphere as a protective layer, absorbing the majority of the sun’s harmful ultraviolet radiation. Ozone is an inorganic molecule also known as oxygen 3, trioxygen or o3. it derives its name from the greek verb ozein, meaning “to smell,” due to its specific odor. its positive or negative effects depend on where it is found. Ozone is an inorganic oxygen molecule with the chemical formula o 3. it is a reactive allotrope of oxygen that also goes by the name trioxygen. ozone protects the planet from harmful ultraviolet radiation, yet is also acts as a pollutant near the earth’s surface and carries health risks. Ozone (o₃) is a reactive form of oxygen found mainly in the atmosphere around 20 km above sea level. it forms from oxygen when exposed to sunlight and is crucial in protecting earth by absorbing harmful ultraviolet (uv) radiation. ozone (o3) is a polar molecule with a bent geometry. Ozone is rarely called trioxygen, even though this is its iupac systematic name. [5] . ozone is formed from oxygen gas (o 2) by the action of ultraviolet light and also atmospheric electrical discharges. it is present in low concentrations throughout the earth's atmosphere.

Nemu Sindi Amora Dts Music Youtube Ozone is an inorganic molecule also known as oxygen 3, trioxygen or o3. it derives its name from the greek verb ozein, meaning “to smell,” due to its specific odor. its positive or negative effects depend on where it is found. Ozone is an inorganic oxygen molecule with the chemical formula o 3. it is a reactive allotrope of oxygen that also goes by the name trioxygen. ozone protects the planet from harmful ultraviolet radiation, yet is also acts as a pollutant near the earth’s surface and carries health risks. Ozone (o₃) is a reactive form of oxygen found mainly in the atmosphere around 20 km above sea level. it forms from oxygen when exposed to sunlight and is crucial in protecting earth by absorbing harmful ultraviolet (uv) radiation. ozone (o3) is a polar molecule with a bent geometry. Ozone is rarely called trioxygen, even though this is its iupac systematic name. [5] . ozone is formed from oxygen gas (o 2) by the action of ultraviolet light and also atmospheric electrical discharges. it is present in low concentrations throughout the earth's atmosphere.
Comments are closed.