2018 Introduction To Electrochemical Cells

Electrochemical Cells Pdf Electrochemistry Redox To obtain a true estimate of the actual open circuit cell voltage, eeq in the fully charged state for operation of the battery; the theoretical cell voltage is modified by the nernst equation, which takes into account the nonstandard state of the reacting component as e eq = e0 − rt ln q (1.2). Pdf | basics of electrochemical cell. | find, read and cite all the research you need on researchgate.

Introduction To Electrochemistry Electrochemical Cells With Anno Pdf This chapter introduces electrochemical cells, specifically batteries, explaining their components, such as electrodes, electrolytes, and separators, along with their functions in storing electrical energy as chemical energy. This document provides an overview of electrochemistry and energy storage systems. it discusses key thermodynamic concepts like internal energy, enthalpy, free energy and their relationships. Scientifi cally batteries are referred to as electrochemical or galvanic cells, due to the fact that they store electrical energy in the form of chemical energy and because the electrochemical reactions that take place are also termed galvanic. Scientifically, batteries are referred to as electrochemical or galvanic cells, due to the fact that they store electrical energy in the form of chemical energy and because the electrochemical reactions that take place are also termed galvanic.

Electrochemical Cells Prompts Stable Diffusion Online Scientifi cally batteries are referred to as electrochemical or galvanic cells, due to the fact that they store electrical energy in the form of chemical energy and because the electrochemical reactions that take place are also termed galvanic. Scientifically, batteries are referred to as electrochemical or galvanic cells, due to the fact that they store electrical energy in the form of chemical energy and because the electrochemical reactions that take place are also termed galvanic. Electrochemical reactions can be conducted with a variety of simple setups including a three necked flask as the reaction vessel, mechanical pencil leads as electrodes, and a phone charger as a power supply. Electrochemical cells a complete cell contains: conducting electrodes (metal, carbon) completed circuit (for electrons to flow) a salt bridge (usually!) an electrolyte solution. This brand new addition to the series provides the most accessible first introduction to electrochemistry, combining explanation of the fundamental concepts with practical examples of how. This simplified relationship is frequently used in electrochemical modeling for fast electrode processes in batteries (e.g., lithium anode) and fuel cells (e.g., hydrogen anode).
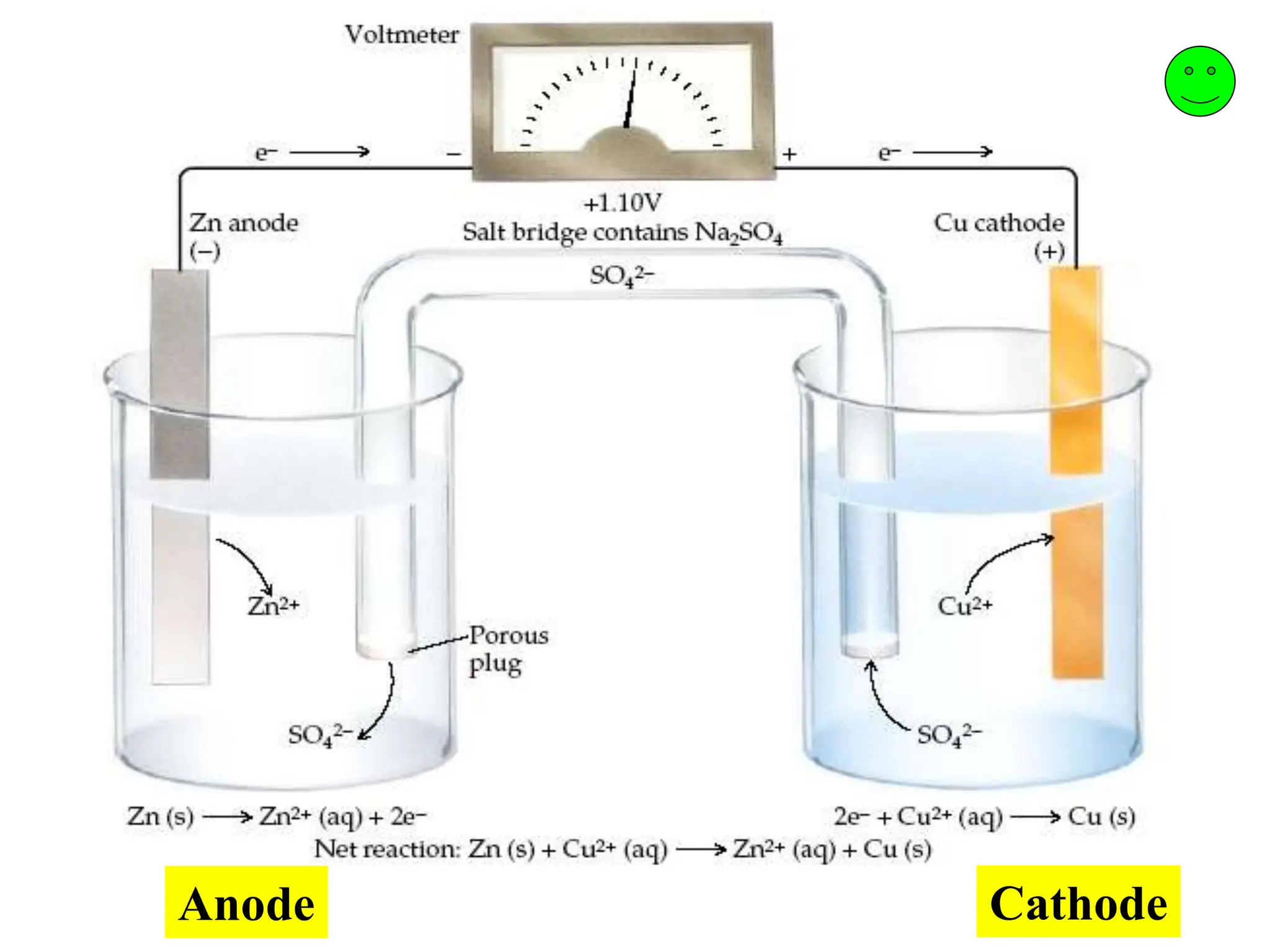
Electrochemistry Introduction Electrochemical Cells Ppt Electrochemical reactions can be conducted with a variety of simple setups including a three necked flask as the reaction vessel, mechanical pencil leads as electrodes, and a phone charger as a power supply. Electrochemical cells a complete cell contains: conducting electrodes (metal, carbon) completed circuit (for electrons to flow) a salt bridge (usually!) an electrolyte solution. This brand new addition to the series provides the most accessible first introduction to electrochemistry, combining explanation of the fundamental concepts with practical examples of how. This simplified relationship is frequently used in electrochemical modeling for fast electrode processes in batteries (e.g., lithium anode) and fuel cells (e.g., hydrogen anode).
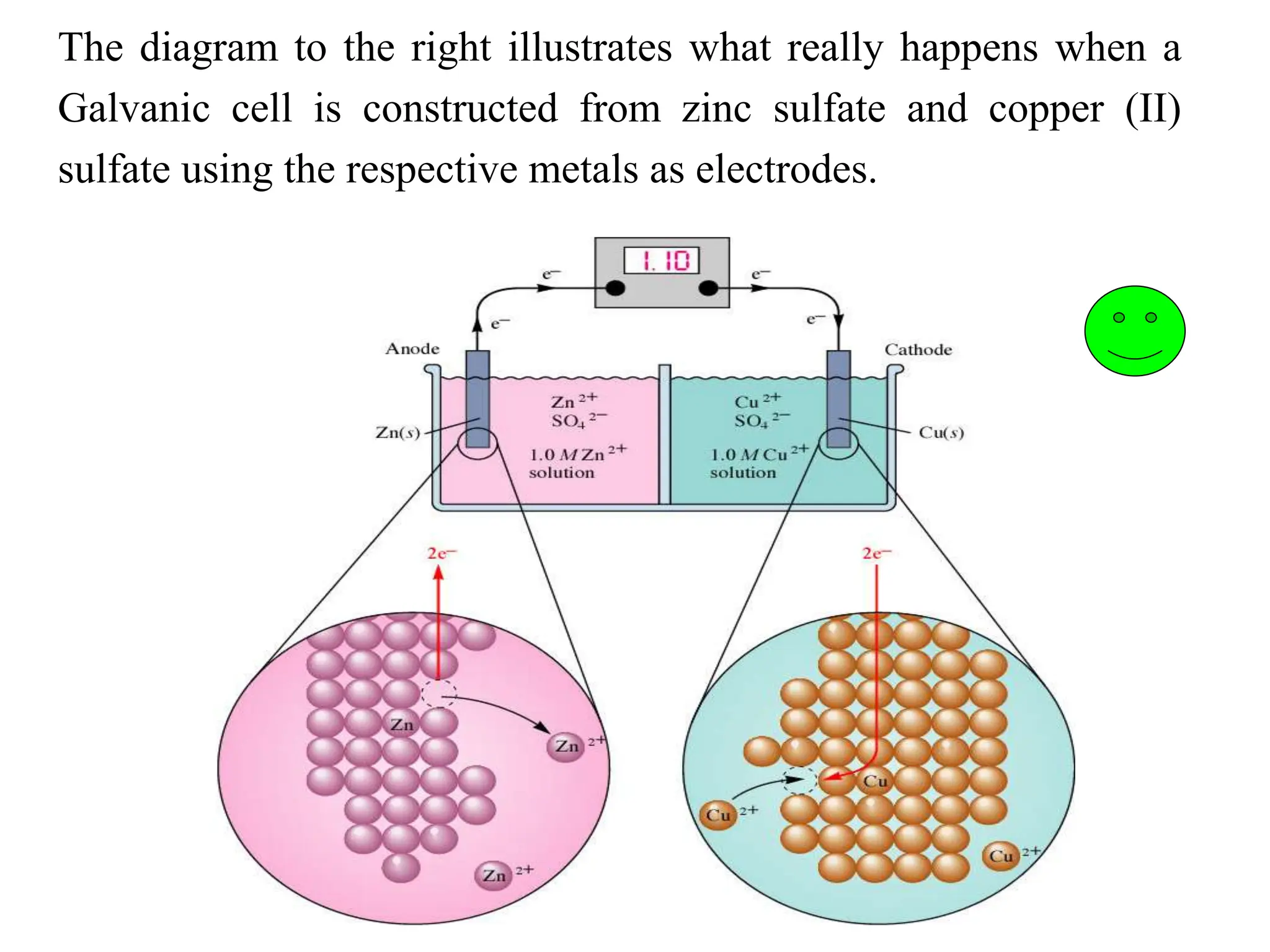
Electrochemistry Introduction Electrochemical Cells Ppt This brand new addition to the series provides the most accessible first introduction to electrochemistry, combining explanation of the fundamental concepts with practical examples of how. This simplified relationship is frequently used in electrochemical modeling for fast electrode processes in batteries (e.g., lithium anode) and fuel cells (e.g., hydrogen anode).
Comments are closed.