20 5 Radial Probability Distribution Function

Radial Probability Distribution Function With Anno Pdf These plots solve the problem posed by the simple “probability distribution curves” which suggested that the probability of finding the electron must be highest at the center of the nucleus in the ground electronic state. In statistical mechanics, the radial distribution function, (or pair correlation function) in a system of particles (atoms, molecules, colloids, etc.), describes how density varies as a function of distance from a reference particle.

Radial Probability Distribution Function Big Chemical Encyclopedia In this article, the rdf was reviewed, including its mathematical formulation, key features, and various methods for its determination, such as x ray and neutron scattering, extended x ray absorption fine structure (exafs), integral equations, and molecular dynamics (md) simulations. The radial distribution function (rdf) defines the probability of finding a particle at distance r from another tagged particle. here, the distance r is between the oxygen atoms of two water molecules. The radial distribution function is most commonly used in gasses, liquids, and solutions, since it can be used to calculate thermodynamic properties such as the internal energy and pressure of the system. Subscribed 13 766 views 4 years ago radial probability distribution function expectation values of radius, potential energy, and kinetic energy more.
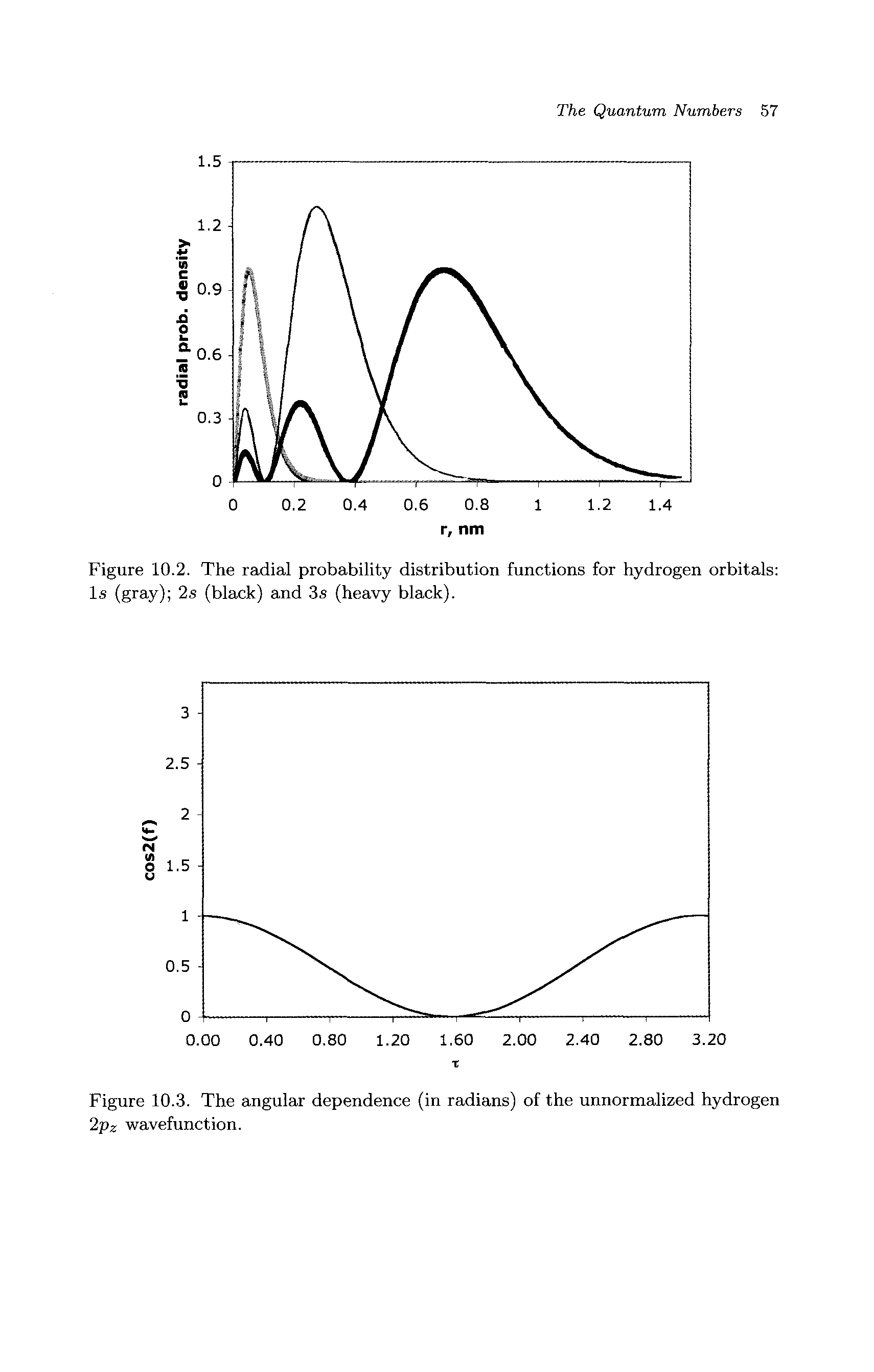
Radial Probability Distribution Function Big Chemical Encyclopedia The radial distribution function is most commonly used in gasses, liquids, and solutions, since it can be used to calculate thermodynamic properties such as the internal energy and pressure of the system. Subscribed 13 766 views 4 years ago radial probability distribution function expectation values of radius, potential energy, and kinetic energy more. It is also known as radial probability density function, it is given by 4πr 2 r 2nl (r). in the graphs shown in question, ψ 2 is shown instead of r 2nl (r). it gives idea about the distribution of electron density at a radial distance around the nucleus without considering the direction or angle. The radial portion of the wavefunction really only tells us if there is high or low probability at various distances from the nucleus (possible radii for the electrons). Compute radial distribution values from particle data for fluids. estimate density, shell volume, and pair statistics. export tables to csv or pdf for reports quickly. It explains how to calculate radial probability, compares different orbitals, and outlines the rules for filling electrons in various orbitals based on the aufbau principle and the (n l) rule.
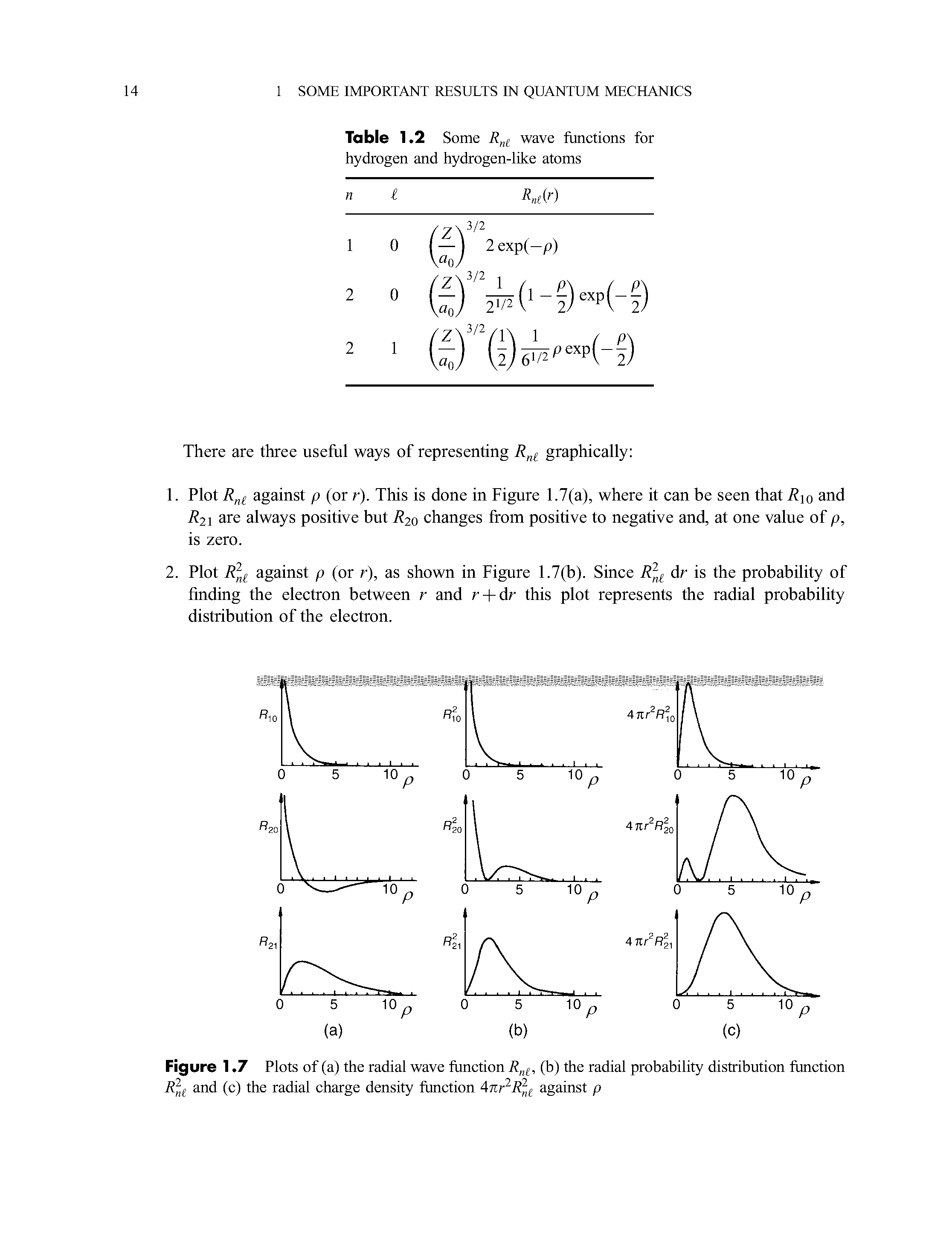
Radial Probability Distribution Function Big Chemical Encyclopedia It is also known as radial probability density function, it is given by 4πr 2 r 2nl (r). in the graphs shown in question, ψ 2 is shown instead of r 2nl (r). it gives idea about the distribution of electron density at a radial distance around the nucleus without considering the direction or angle. The radial portion of the wavefunction really only tells us if there is high or low probability at various distances from the nucleus (possible radii for the electrons). Compute radial distribution values from particle data for fluids. estimate density, shell volume, and pair statistics. export tables to csv or pdf for reports quickly. It explains how to calculate radial probability, compares different orbitals, and outlines the rules for filling electrons in various orbitals based on the aufbau principle and the (n l) rule.

Radial Probability Distribution Function Big Chemical Encyclopedia Compute radial distribution values from particle data for fluids. estimate density, shell volume, and pair statistics. export tables to csv or pdf for reports quickly. It explains how to calculate radial probability, compares different orbitals, and outlines the rules for filling electrons in various orbitals based on the aufbau principle and the (n l) rule.

Consider The Radial Probability Distribution Function Given Below 0 п в ќ
Comments are closed.