1b6 Diatomic Elements
Diatomic Elements Science Trends Diatomic elements are pure elements that form molecules consisting of two atoms bonded together. there are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, and bromine. Some elements cannot exist by themselves, even when they are isolated from any other type of atom. elements of this nature will combine with atoms of the same element in order to be stable. in other words, hydrogen, a diatomic element, cannot be by itself.

What Are The 7 Diatomic Elements Definition And List While some elements exist as molecules, most elements exist with individual atoms as their basic unit. if an element is not one of those listed above as a molecular element, assume it is an atomic element. The periodic table has several diatomic elements, sometimes known as “molecular elements”. let’s learn what they are, and how they are different from diatomic molecules. Interactive periodic table showing names, electrons, and oxidation states. visualize trends, 3d orbitals, isotopes, and mix compounds. fully descriptive writeups. In representing a diatomic molecule, we use the symbol of the element and include the subscript 2 to indicate that two atoms of that element are joined together.
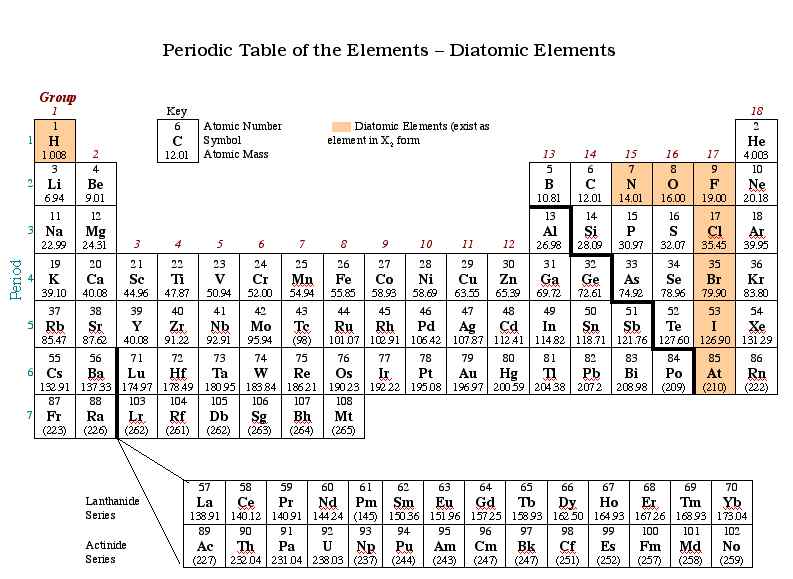
1b6 Diatomic Elements Interactive periodic table showing names, electrons, and oxidation states. visualize trends, 3d orbitals, isotopes, and mix compounds. fully descriptive writeups. In representing a diatomic molecule, we use the symbol of the element and include the subscript 2 to indicate that two atoms of that element are joined together. The diatomic elements include hydrogen (h), nitrogen (n), oxygen (o), fluorine (f), chlorine (cl), bromine (br), and iodine (i). in nature, the diatomic elements only exist as h2, n2, o2, f2, cl2, br2, and i2. The seven diatomic elements are hydrogen (h), nitrogen (n), oxygen (o), fluorine (f), chlorine (cl), bromine (br), and iodine (i). these seven elements are found in various locations on the periodic table. The periodic table has several diatomic elements, sometimes known as “molecular elements”. let’s learn what they are, and how they are different from diatomic molecules. We are constantly referring to the elements on the periodic table. seven of those elements exist as diatomic molecules meaning the formula is not just the symbol by itself, but the symbol subscript 2.
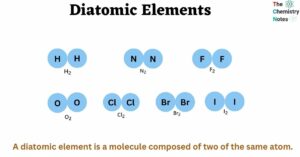
Diatomic Elements Important 7 Elements Properties Formation The diatomic elements include hydrogen (h), nitrogen (n), oxygen (o), fluorine (f), chlorine (cl), bromine (br), and iodine (i). in nature, the diatomic elements only exist as h2, n2, o2, f2, cl2, br2, and i2. The seven diatomic elements are hydrogen (h), nitrogen (n), oxygen (o), fluorine (f), chlorine (cl), bromine (br), and iodine (i). these seven elements are found in various locations on the periodic table. The periodic table has several diatomic elements, sometimes known as “molecular elements”. let’s learn what they are, and how they are different from diatomic molecules. We are constantly referring to the elements on the periodic table. seven of those elements exist as diatomic molecules meaning the formula is not just the symbol by itself, but the symbol subscript 2.
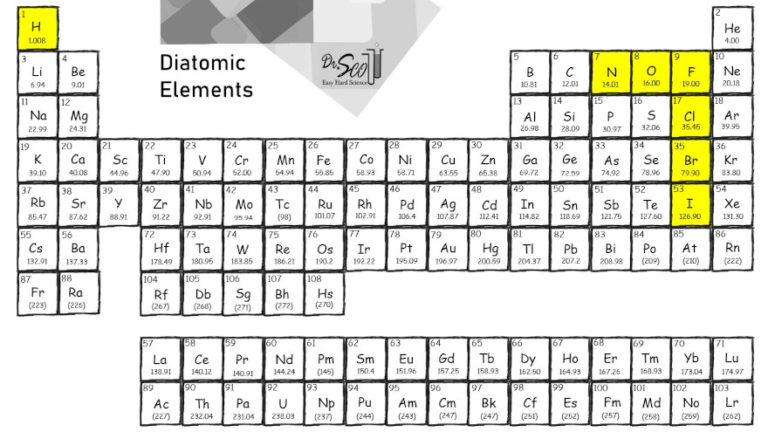
Diatomic Elements Easy Hard Science The periodic table has several diatomic elements, sometimes known as “molecular elements”. let’s learn what they are, and how they are different from diatomic molecules. We are constantly referring to the elements on the periodic table. seven of those elements exist as diatomic molecules meaning the formula is not just the symbol by itself, but the symbol subscript 2.
Comments are closed.